
CNC Turned Parts for Medical Devices – Precision That Saves Lives
Date:2026-04-16Article editor:Starting Point PrecisionViews:65The medical device industry demands absolute reliability. Every component, no matter how small, must perform flawlessly inside the human body or in critical surgical tools. CNC turned parts for medical devices are manufactured to tolerances as tight as ±0.005 mm, using biocompatible materials and validated processes. This article explains why precision turning is essential for medical applications and how to choose a trusted supplier that meets regulatory standards.
What Are CNC Turned Parts in Medical Manufacturing?
CNC turning uses computer-controlled lathes to rotate a workpiece while a stationary cutting tool removes material. This process produces cylindrical, conical, or complex contoured parts such as bone screws, biopsy needle hubs, dental implant abutments, and surgical instrument handles. For medical devices, the requirements go beyond geometry – material traceability, surface finish (Ra ≤ 0.2 µm), and contamination control are mandatory.
Key Medical Applications of CNC Turned Parts
Medical Field | Typical Turned Components |
| Orthopedics | Bone screws, locking caps, trial spacers |
| Cardiovascular | Guidewire mandrels, catheter connectors, stent crimping dies |
Dental | Implant healing abutments, transfer copings |
Surgical Instruments | Scissor screws, trocar tips, endoscope adapters |
Drug Delivery | Pen injector bodies, vial adapter spikes |
Critical Requirements for Medical-Grade CNC Turning
1. Biocompatible Materials
Only certified materials such as stainless steel (316L, 17-4PH), titanium (Grade 5 Ti-6Al-4V), PEEK, and medical-grade brass are used. Each batch must come with an EN 10204 3.1 material certificate.
2. Cleanliness and Sterilization Compatibility
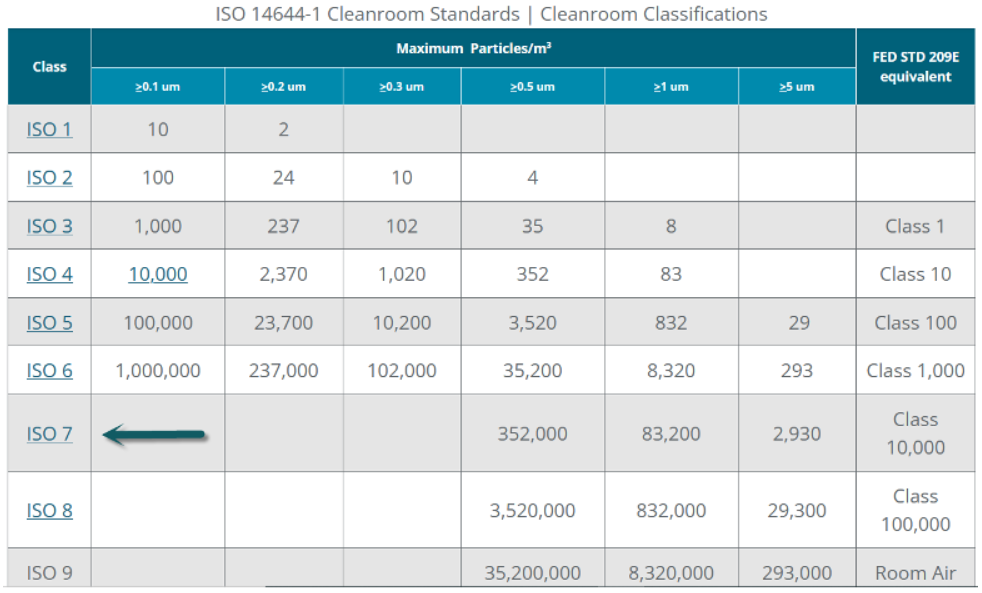
Parts must be free from machining oil, burrs, and particulate contamination. Many components require ultrasonic cleaning, passivation (for stainless steel), or anodizing (for titanium) before final packaging in cleanroom conditions (ISO Class 7 or better).
3. Quality Management Systems
Suppliers should hold ISO 13485:2016 certification (medical devices quality management) and comply with FDA 21 CFR Part 820 for devices sold in the US. CE marking (MDR 2017/745) is required for European markets.

How CNC Turning Achieves Medical-Grade Precision
Modern Swiss-type CNC lathes with live tooling can produce parts up to 32 mm in diameter with:
● Tolerances: ±0.005 mm for critical features
● Surface finish: Ra 0.1–0.2 µm without secondary polishing
● Complexity: Threads, undercuts, cross holes, and polygonal profiles in one setup
We use in-process probing and after-process vision inspection (CMM) to guarantee 100% dimensional compliance. A typical medical part – a titanium bone screw – is shown below:
CNC turned titanium bone screw for spinal fusion (length 28 mm, thread pitch 0.8 mm)
Regulatory Compliance and Traceability
Every supplier must demonstrate:
● Experience: Minimum 5 years supplying Class II/III medical components.
● Expertise: Validated process capability (Cpk ≥ 1.33) and documented risk management per ISO 14971.
● Authoritativeness: ISO 13485 certification and FDA establishment registration.
● Trustworthiness: Full lot traceability from raw material to finished part, including all machining parameters and inspection records.
How to Validate a Supplier’s Medical Capabilities
Before placing an order, request:
1. ISO 13485 certificate (current, with notified body stamp).
2. A validated process flow chart including cleaning and packaging.
3. Material certifications for the exact lot you intend to purchase.
4. First article inspection report (FAIR) per AS9102 or customer specification.
We provide all four documents with every initial sample order. To start your project, submit your drawing via request-quote.
Conclusion
CNC turned parts for medical devices require more than just machining skills – they demand a culture of quality, cleanliness, and regulatory discipline. Partner with a supplier that holds ISO 13485, uses validated processes, and provides full traceability. Your patients’ safety depends on it.




Copyright © 2019 All Rights Reserved Dongguan Start Precision Technology Co., Ltd. Tel: +86-769-82855591
Add: No. 277 Zhen'an Middle Road, Chang'an Town, Dongguan, Guangdong, China