
ISO 13485 Certification for Medical CNC Machining Parts: Ensuring Quality and Compliance
Date:2026-03-19Article editor:Starting Point PrecisionViews:212The medical device industry operates under some of the most stringent regulations in the world. Components manufactured through Computer Numerical Control (CNC) machining—such as orthopaedic implants, surgical instruments, and dental prostheses—must exhibit flawless precision and absolute reliability. To meet these demands, manufacturers increasingly pursue ISO 13485 certification. This internationally recognized standard specifies requirements for a quality management system (QMS) specific to medical devices. For companies producing medical CNC machining parts, ISO 13485 is more than a certificate; it is a gateway to global markets and a testament to patient safety.
What is ISO 13485?
ISO 13485 is a stand-alone standard, based on the ISO 9001 framework but tailored for the medical device sector. It defines the requirements for a QMS that enables an organization to consistently provide medical devices and related services that meet customer and applicable regulatory requirements. Unlike general quality standards, ISO 13485 places heightened emphasis on risk management, design controls, process validation, and traceability—all critical factors when machining parts that will be implanted in the human body or used in life-saving procedures.
Why Medical CNC Machining Parts Need ISO 13485
CNC machining produces high-precision components from materials like titanium, stainless steel, and advanced polymers. These parts often contact sterile fields or remain inside the body for years. A single dimensional error, surface defect, or contamination can lead to device failure, patient harm, and manufacturer liability. Regulatory authorities such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency require medical device manufacturers to implement a QMS that aligns with ISO 13485 (or the U.S. equivalent, 21 CFR 820). Certification demonstrates that a CNC machining supplier has robust processes for:
● Process validation (e.g., machining parameters, cleaning, packaging)
● Supplier management and incoming material control
● Traceability from raw material to finished part
● Handling non‑conformities and implementing corrective actions
Moreover, ISO 13485 facilitates smoother regulatory approvals and market access worldwide. Many medical device OEMs mandate that their CNC machining partners hold current certification before entering supply agreements.
Key Requirements of ISO 13485 for CNC Machining
Implementing ISO 13485 in a CNC machining environment involves several specific elements:
1. Document Control and Records – Every procedure, work instruction, and quality record must be meticulously managed and retained.
2. Management Responsibility – Top management must demonstrate commitment by establishing a quality policy, conducting management reviews, and ensuring resource availability.
3. Resource Management – Personnel must be competent, and the work environment (e.g., cleanliness, temperature control) must be suitable for medical device production.
4. Product Realization – This includes planning, customer-related processes (contract review), purchasing control, and production validation. For CNC machining, special attention is given to equipment maintenance, process validation (e.g., for critical dimensions or cleaning), and identification/traceability.
5. Measurement, Analysis, and Improvement – Monitoring of processes, internal audits, handling of non‑conforming product, and implementation of corrective/preventive actions are mandatory.
6. Risk Management – Following ISO 14971. risks associated with the manufacturing process and the final part must be assessed and mitigated.
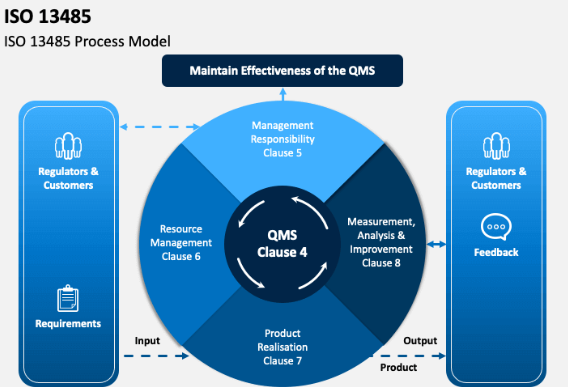
ISO 13485 QMS model showing continuous improvement cycle
Benefits of Certification
Achieving ISO 13485 certification brings tangible advantages:
● Customer Confidence – OEMs prefer certified suppliers because it reduces their audit burden and assures compliance.
● Regulatory Alignment – Certification helps meet FDA, MDR, and other international requirements, streamlining product approvals.
● Operational Excellence – The disciplined QMS reduces scrap, rework, and variability, leading to cost savings.
● Market Differentiation – In a competitive landscape, ISO 13485 sets a supplier apart as a quality-focused partner.
For an authoritative overview of the standard, visit official ISO 13485 page.
Steps to Achieve ISO 13485 Certification
The path to certification typically follows these stages:
1. Gap Analysis – Compare current practices against ISO 13485 requirements.
2. Documentation – Develop a quality manual, procedures, and work instructions tailored to CNC machining.
3. Implementation – Train employees, execute the QMS, and collect records.
4. Internal Audit – Verify that the system functions as intended.
5. Management Review – Evaluate QMS performance and identify improvements.
6. Certification Audit – An accredited body conducts a two‑stage audit; any non‑conformities must be resolved.
7. Surveillance – Annual audits maintain the certificate.
Conclusion
ISO 13485 certification is not a one‑time achievement but a continuous commitment to quality and patient safety. For manufacturers of medical CNC machining parts, it provides the framework to consistently deliver components that meet the highest standards. In an industry where precision can mean the difference between life and death, ISO 13485 is the benchmark that earns trust and drives excellence.





Copyright © 2019 All Rights Reserved Dongguan Start Precision Technology Co., Ltd. Tel: +86-769-82855591
Add: No. 277 Zhen'an Middle Road, Chang'an Town, Dongguan, Guangdong, China